The following statements were made by Patrick Roddie in a statement he read at an EPA public comment hearing on greenhouse gases in aircraft exhaust. Roddie is a proponent of the "chemtrail" theory, thinking that persistent contrails are actually something toxic being secretly sprayed by the US government. Roddie was recently featured in an SF Weekly article on the chemtrail theory.
http://stopsprayingus-sf.com/epa/
Now the first paragraph here is true. Those are the refractive indices of liquid water and ice. Rainbows do form at a radius of 42 degrees. Rainbows are centered on the antisolar point (the point opposite the sun).
Water and ice have refractive indices of 1.333 and 1.309 respectively and produce rainbows with an angular radius of 42 degrees, centered on the antisolar point.
But in recent years a formerly rare phenomenon has become commonplace, a 21 degree halo completely encircling the sun. Some argue that these halos or “incredibly rare sun dogs” are formed by ice crystals, but nothing can change the refractive index of water or ice - which form 42 degree halos. Metal salts have a higher refractive index and therefore form much tighter halos. Crystalline aluminum oxide, for example, has a refractive index of 1.762-1.778 while barium sulfate has a refractive index of 1.636.
My contention that these “incredibly rare sun dogs” are formed by metal salts with a higher refractive index than water is reinforced by rainwater analysis taken during a 30 day period when I recorded 21 of these halos in March/April 2015. I collected rainwater in clean glass bowls on the roof of my San Francisco apartment building on April 5th, 2015, six thousand miles downwind from the nearest factory, power plant, refinery, freeway, quarry or mine. I sent it to a NELAP certified lab and they recorded barium at a staggering 160 micrograms per liter. Less than a gram will kill an adult human.
But the key point of the next paragraph, that water or ice is constrained by its refractive index to form 42 degree halos, is almost entirely wrong. Rainbows are not halos.
A halo is a ring that forms around the sun. A rainbow is a ring (of which you normally only see the top) that forms opposite the sun. A halo is made from the sunlight refracting through hexagonal ice crystals. A rainbow is formed by the sun refracting and reflecting though spherical drops of liquid water. These are two very different things.
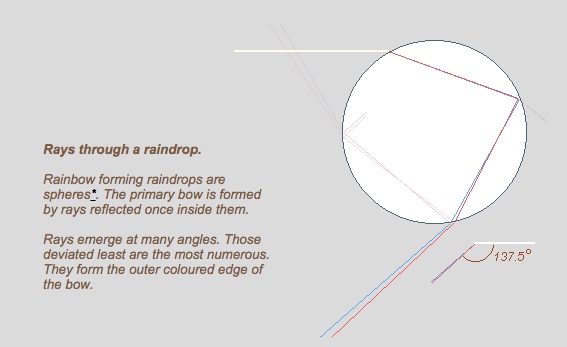
Both rainbows and halos are made by light scattering in multiple directions. The direction in which they are scattered the least forms the inner radius of the rainbow or halo. This is known as the "angle of minimum deviation".
As rainbow actually refract and reflect the light back towards the sun, the angle of minimum deviation is large, 137.5, but we measure the radius from the centerline, so it's 180-137.5 or 42.5. Commonly rounded to 42 degrees.
http://www.atoptics.co.uk/rainbows/primrays.htm
Halos just refract the light on the way from the sun. Again they refract though many angles, but the angle of minimum deviation is 21.7°, this is commonly rounded to 22°
http://www.atoptics.co.uk/halo/circ1.htm
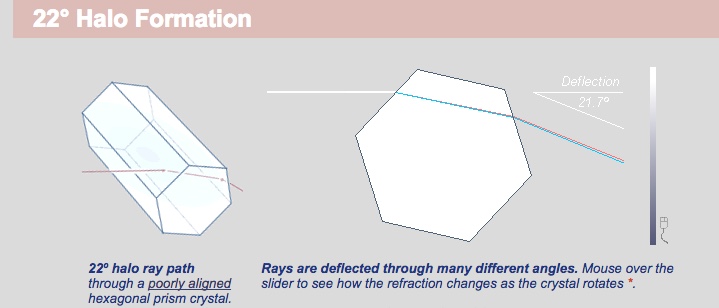
The links above, to the excellent Atmospheric Optics web site, have interactive versions of the above diagrams, showing the range of possible deviations. It also has many descriptions of different types of atmospheric phenomena.
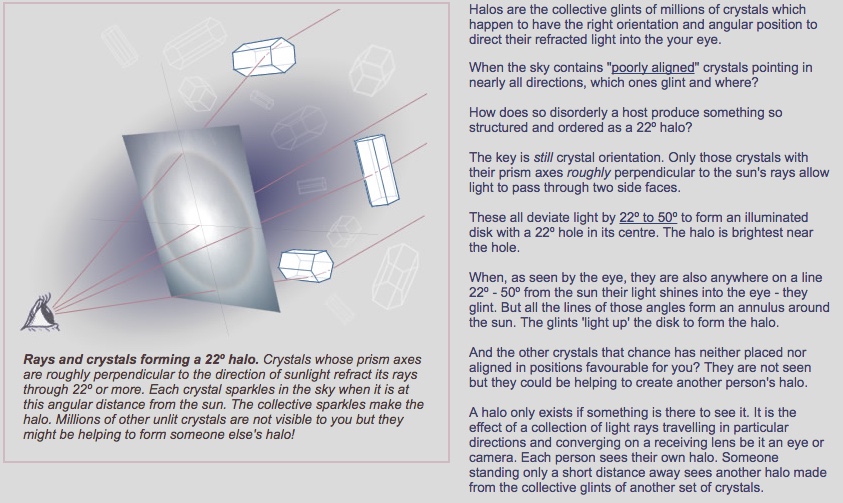
22° halos are not at all a new thing. They have been commonly described in books on the weather, and observed by people since ancient times. They were discussed by Aristotle, 2400 years ago. And back in 1904 22° halos were described in the book Cloud Studies as being "common in most parts of the world."

Halos are not particularly rare. In fact, depending on location, they can occur more frequently than rainbows. See the Encyclopedia Brittanica, 1823 (note they use the older general term "corona"
https://books.google.com/books?id=v...nepage&q=encyclopedia britannica halo&f=false

Slightly more rare is the sun dog - which is also constrained by geometry to appear at 22° (as it appears on the sides the halo). But again, in no sense is it incredibly rare - although it does vary by location. Anecdotally I see a lot more sun-dogs and halos in Sacramento than I did in Los Angeles.
But what of the claim that "21° halos" are evidence of being formed by some chemical with a higher refractive index than water? This is kind of a moot point, as we now know that 21.7° (generally rounded up to 22°, not down to 21°) is exactly the size of halo you would expect from the basic geometry of ice crystals, and is the type that has been observed for thousands of years. But what if we used something with a higher refractive index?
As you'd imagine, a higher refractive index causes light to bend more. Since it bends more, the minimum deviation angle is larger. So the inner radius of the the halo would be bigger.
Remember, Roddie claimed "Metal salts have a higher refractive index and therefore form much tighter halos.", and yet the opposite is true. With a higher refractive index, the halo would be bigger.
So, seeing a 22° halo is not only not evidence that something other than water is creating that halo, it's actually absolute proof that whatever is creating the halo has the same refractive index of water ice (1.309) (which of course suggest the most likely explanation is just water ice). So it can't be, for example, the two things that Roddie suggest: Crystalline aluminum oxide(1.762-1.778). or barium sulfate (1.636), as they would create much bigger halos.
Bigger halos of different types do exist, but like the common 22° halos, they are all at the exact position you would expect for hexagonal ice crystals. There's even a very rare 46° halo, generally only seen in very cold regions.
http://www.atoptics.co.uk/halo/46hal.htm

But one thing there isn't is a 42° halo, despite Roddie's statement "nothing can change the refractive index of water or ice - which form 42 degree halos" - that's just flat wrong.
Finally, one more moot point, as there's no evidence of barium sulfate being sprayed, but Roddie states "Less than a gram will kill an adult human." However barium sulfate is almost entirely non-toxic. In fact it's so inert that hospitals make smoothies out of barium sulphate to use as an x-ray visibility agent. Each one contains several grams of barium sulphate (9 grams in the common Readi-Cat 2).

Of course there are also toxic compounds of barium, but Roddie only refers to spraying barium sulfate.
To sum up: Normal common halos and sun dogs are at 22°, and always have been. If they were not at 22° then that would mean they were being formed by something other than ice. They are only observed at 22°. So they can't be made of aluminum oxide, or barium sulfate, which isn't even toxic.
Roddie is very wrong here, and it's not the only thing he's wrong about in his statement. If this is the evidence his theory is based on, then I suggest he should retract his EPA statement - or at the very least issue an errata.
--------------------------------------------------------------------------------------------
For more discussion of Halos, see:
https://www.metabunk.org/debunked-chemtrail-chembows-solar-halos.t3916/
Last edited: