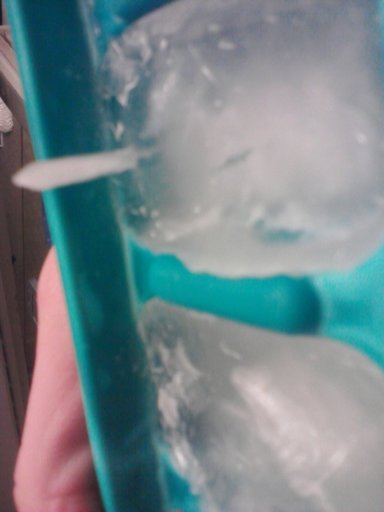
Sometimes when you make ice cubes in the freezer, you get odd spikes on top of the ice:

This does not happen all the time, and many people will never see this. It depends on what's in your water, the temperature of your freezer, and if there's a fan in the freezer. They form most frequently with distilled water, and not very often with regular tap water.
Caltech's SnowCrystals.com page has an explanation as to why they form:
http://www.its.caltech.edu/~atomic/snowcrystals/icespikes/icespikes.htm
Fascinating stuff. But what really got me interested was the claim made at Orgone Innovations (a site that sells "personal orgone generators", aka POGs), that you could create these stalagmites simply by placing a "POG" on top of the freezer. Note that the POG is not some kind of machine. It sounds a bit like an ozone generator, but really it's just a bunch of cheap beads, crystals and wire encased in resin - a pretty paperweight. And it's not place IN the freezer, where you might think it could have an effect, but instead is placed OUTSIDE the freezer, where any effect is going to be something entirely new to science.
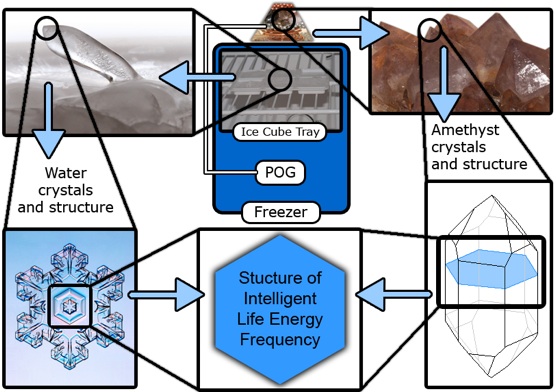
Here's their explanation and schematic of what they think is going on. Compare to the one above:
http://orgoneinnovations.com/?q=ice-stalagmites
mod add: cached version https://web.archive.org/web/20120503204828/http://orgoneinnovations.com/?q=ice-stalagmites
Have I tried it, to verify it does not work? No.
So how can I claim it does not work, if I've never tried it? I know it does not work because nobody has ever demonstrated that it works. If it did work, then it would revolutionize physics. It's a discovery more than worthy of a Nobel Prize. But nobody has ever demonstrated that it works. Sure, people have put distilled water in a freezer, and put the POG on top, and two hours later stalagmites formed, but they would have formed anyway - it's distilled water!. They need to take the extra step of trying it without the POG, and verifying that nothing happened, then with the POG again. They need to do this about ten times both with and without the POG, using the same tray and water. Then document what happens, and collect your Nobel prize.

This does not happen all the time, and many people will never see this. It depends on what's in your water, the temperature of your freezer, and if there's a fan in the freezer. They form most frequently with distilled water, and not very often with regular tap water.
Caltech's SnowCrystals.com page has an explanation as to why they form:
http://www.its.caltech.edu/~atomic/snowcrystals/icespikes/icespikes.htm
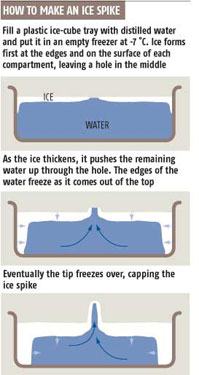
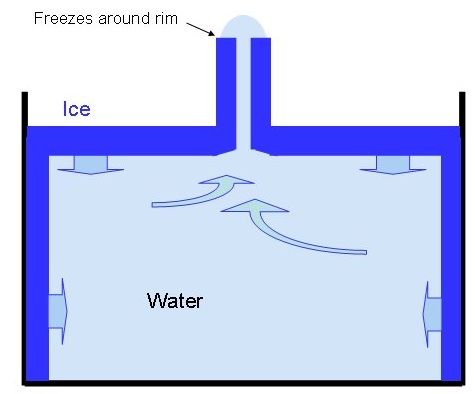
And here's a schematic diagram:Ice spikes grow as the water in an ice cube tray turns to ice. The water first freezes on the top surface, around the edges of what will become the ice cube. The ice slowly freezes in from the edges, until just a small hole is left unfrozen in the surface. At the same time, while the surface is freezing, more ice starts to form around the sides of the cube.
Since ice expands as it freezes, the ice freezing below the surface starts to push water up through the hole in the surface ice (see diagram). If the conditions are just right, then water will be forced out of the hole in the ice and it will freeze into an ice spike, a bit like lava pouring out of a hole in the ground to makes a volcano. But water does not flow down the sides of a thin spike, so in that way it is different from a volcano. Rather, the water freezes around the rim of the tube, and thus adds to its length. The spike can continue growing taller until all the water freezes, cutting off the supply, or until the tube freezes shut.

Fascinating stuff. But what really got me interested was the claim made at Orgone Innovations (a site that sells "personal orgone generators", aka POGs), that you could create these stalagmites simply by placing a "POG" on top of the freezer. Note that the POG is not some kind of machine. It sounds a bit like an ozone generator, but really it's just a bunch of cheap beads, crystals and wire encased in resin - a pretty paperweight. And it's not place IN the freezer, where you might think it could have an effect, but instead is placed OUTSIDE the freezer, where any effect is going to be something entirely new to science.
Here's their explanation and schematic of what they think is going on. Compare to the one above:
http://orgoneinnovations.com/?q=ice-stalagmites
mod add: cached version https://web.archive.org/web/20120503204828/http://orgoneinnovations.com/?q=ice-stalagmites

Does it work? No.
What this simple experiment does is reveal the Positive Orgone Generators effect upon the structure of water. It also shows the permeating nature of Orgone Energy and its ability to transcend obstacles such as insulation, plastic, electrical wiring, metal, paint, and glass. This is easy to understand when we recognize that all matter is energy, and all energy vibrates and changes frequency. Probably the most profound aspect of this experiment is that it shows how greatly Positive Orgone Generators effect water in an intelligent and living manner. This has extreme significance when we recognize that all living things are made from water. Keeping a Positive Orgone Generator with you at all times will charge the water within your body, much like the intelligent/life energy of the ice crystals.
Have I tried it, to verify it does not work? No.
So how can I claim it does not work, if I've never tried it? I know it does not work because nobody has ever demonstrated that it works. If it did work, then it would revolutionize physics. It's a discovery more than worthy of a Nobel Prize. But nobody has ever demonstrated that it works. Sure, people have put distilled water in a freezer, and put the POG on top, and two hours later stalagmites formed, but they would have formed anyway - it's distilled water!. They need to take the extra step of trying it without the POG, and verifying that nothing happened, then with the POG again. They need to do this about ten times both with and without the POG, using the same tray and water. Then document what happens, and collect your Nobel prize.